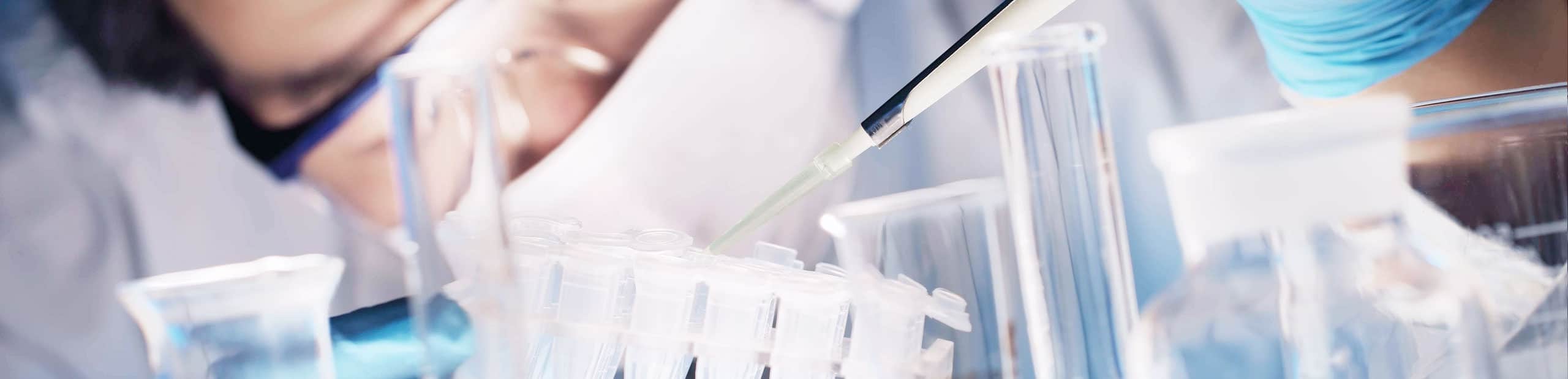
Precision in the clean room. High-quality, customised plastic parts and systems for the medical industry.
As long-term partners to the medical industry, we know that accuracy, process safety and hygiene all save lives on a daily basis. Therefore, we leave nothing to chance.
Constantly increasing demands require new higher-performing technologies and reliable partnerships. This means that high-tech medical components must satisfy the highest cleanliness, precision and process stability specifications throughout the product's entire life and often under extremely sensitive conditions.
Thanks to the experience we have gained from more than 1000 medical engineering projects we are able to bring plastic parts, modules and entire systems both flexibly and precisely to production maturity. We can map tolerances in the µ range and ensure that the quality remains reproducible in every batch. We are happy to advise throughout the entire value-added chain, to inspect and optimise in our own measuring laboratory and to document the entire process. In doing so, we consistently rely on the certification of our quality standards, clean room manufacturing (class 8) and the automation of our manufacturing processes.
- Development partner for the medical technology industry
- 80 years of competence in processing technical plastics
- Clean room manufacturing (class 8)
- Certified according to ISO 13485, and others
- Production-accompanied measurement in own measuring centre
- Very high process stability
- 100 % traceability
For every technical medical requirement, there is a suitable plastic and customer-specific solution. Development partners for more than 80 years.
Together with our customers, we have already launched numerous ideas and guided them to production maturity. This includes not only new product innovations and improvements to existing customer products but also the market introduction of our own patented products.
simplix – our patented in-house product for extracting laboratory samples
A system solution for an economically efficient handling of laboratory samples
The simplix® sample extraction device is a sophisticated and intelligent disposable, which focuses on the demands and needs of the users and together with our customer is continually optimized. Due to its leak-proof, customized color, simple handling and patented locking system, simplix® has already proven itself by being sold millionfold.
simplix® is a reliable and particularly economical solution when it comes to the preparation of samples in an exact volume – contact us today and request your free samples.
Intelligent, functional disposable products
Maximum safety after 100 percent checked quality
A pronounced example for our capabilities in the field of disposables is the Qube® cassette of the Ruck GmbH, which are used in the eponymous Eye-Surgery-System Qube® and Qube® pro. A complete system, which is injection moulded in-house, assembled, ultrasonic welded and 100 percent leak tested. In a following assembly step, hoses and collection bags are added and in the final step, the system is wrapped, packed and ETO sterilized.
The cassette functions as a disposable product and is used together with the Eye-Surgery-System for surgery in the front and back eye segment. The function of the cassette is the fluid supply in the eye (irrigation) as well as the drainage of the liquid and the tissue (aspiration) out of the eye. The density of the cassette plays a key role during this procedure, in order to build up the necessary vacuum for the surgery.
Visual and functional parts for treatment units
Fulfilling top requirements regarding optics, cleaning properties and chemical resistance
We manufacture a complete range of parts and modules for the treatment unit. Priority is given to achieving highly polished surface structures that facilitate an optimum disinfection and improve the resistance of the plastic used against aggressive chemicals and substances.
- Amalgam rotor
- Bite-down element
- Instrument tray
- Beaker outlet
- Housing
Complete housing for blood analysis devices
A focus on accuracy of fit, haptic and reliability
We manufacture a large range of parts, modules and systems for blood sugar and blood coagulation measuring devices. By overmoulding the inserts using injection moulding, we are able to reduce the number of process steps and implement 2-component technology with differing haptics and surfaces in a single cycle. One of our particular strengths in this area is the numerous assembly and post-processing options, to customised final packaging of the entire module:
- Heat staking
- Pad printing
- Ultrasound welding of plastic parts
- Use of purchased parts such as displays, electronics and boards
- Screw connections
- Gluing in and embedding of metal parts and magnets
- Module packaging for end customers
The following well-known customers have been trusting in our capability for many years:
Fulfilment of top hygiene and cleanliness specifications – GAUDLITZ clean room manufacturing (class 8).
In our clean room, we process almost all usual thermoplastics using clean room suitable injection moulding machines under controlled and standardised conditions. As well as providing customer-specific automation, we can assemble complex modules and handle all packaging needs. In doing so, we meet the highest cleanliness and dust-free specifications in a particle-reduced environment.
- Clean room manufacturing (class 8)
- Clean room suitable automation systems
- Assembly and processing under clean room conditions
Certified manufacturing for consistent processes. This is how we ensure your level of quality.
We rely on the consistent certification of our quality standard, clear standard operating procedures as part of our integrated management system, product steering plans and one-hundred-percent traceability to the EOP. In order to ensure that
the quality remains consistent, we analyse, measure and optimise components and tool components in our GAUDLITZ measuring center – from the industrial measuring procedure to non-destructive computed tomography. We test the substitution using alternative materials, carry out preventive feasibility analyses and take on the customer-specific design of tools and automation processes.
- ISO TS 16949
- ISO 14001
- ISO 9001
- ISO 13485
- ISO 50001
- AEO certificate
Do you have a great idea? We can implement it. Your one-stop partners for development, tool engineering, plastics processing and system assembly.
Benefit from the advantages of having an experienced system supplier.
On site, we have all the requisite materials expertise, injection moulding technology, metrology equipment, available capacity and clean room conditions required for an efficient and economical implementation of the complete development and manufacturing process for any batch size.
Our strengths lie in high-tech materials and process consultation, the plastics-compatible design of tools and components and the manufacture of extremely precise and customer-specific plastic products. Thanks to our assembly and post-processing facilities, we can offer you the entire process under one roof.
Put us to the test and contact us.
Send your enquiry to us by E-mail or leave a message so that we can call you back. Our Sales Management Team will be in touch with you as soon as possible.